Precision Medical Robotics for Radiotherapy & Surgery
Certified robotic systems for radiotherapy, particle therapy and surgical assistance, developed with leading clinical centres and built to medical device standards.
Explore Exacure Contact Medical Team
Built to Medical Device Standards
BEC medical systems are developed and manufactured in compliance with applicable medical device regulations. Every system undergoes rigorous validation before clinical deployment.
Product Card
List and describe the key features of your solution or service.

Exacure
Modular robotic patient positioning system for radiotherapy, particle therapy and BNCT. CE-marked and clinically validated at leading treatment centres in Europe and the USA.
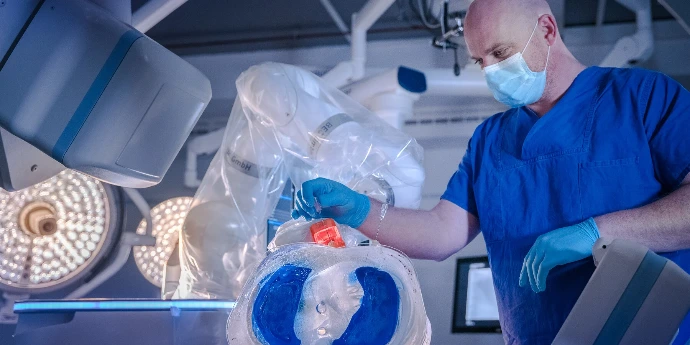
Guidoo
Robotic assistance system for percutaneous biopsies and ablative procedures. Guides needle placement with robot precision. First-in-Human study initiated. CE approval in progress.
Projects
Check out what's new in our company !
Frequently asked questions
Here are some common questions about our company.
BNCT is a cancer treatment that uses a neutron beam to activate boron compounds pre-loaded into tumour cells, destroying them from within while minimising damage to surrounding tissue. It is particularly effective for tumours in locations that are difficult to treat with conventional radiotherapy or surgery. BEC's Exacure system provides the robotic patient positioning required for accurate BNCT beam delivery.
Guidoo is currently progressing through the CE approval pathway and is not yet commercially approved for clinical use. A First-in-Human study has been initiated. BEC provides transparent status updates to clinical partners. Please contact us for the current regulatory status.
Exacure supports BNCT, conventional radiotherapy and particle therapy including proton and carbon ion treatment. The modular system architecture allows it to be configured for different treatment room layouts and neutron source types, including compact accelerator-based neutron sources.
Exacure holds CE Mark approval under Medical Device Regulation, certified by Notified Body CE0483. The system is developed under an ISO 13485 certified quality management system. It is also the first patient positioning system certified to meet the specific requirements of BNCT treatment.
Contact Form
Use this section to boost your company's credibility.
Speak to BEC's Medical Team.
Whether you are planning a new treatment facility, evaluating patient positioning systems, or researching robotic surgical assistance, our medical team is available to discuss your requirements in detail.



