Guidoo — Robotic Assistance System for Percutaneous Biopsies & Ablative Procedures'
KUKA Innovation Award Finalist 2025. First-in-Human study initiated
Request Clinical Information Read the Clinical Study
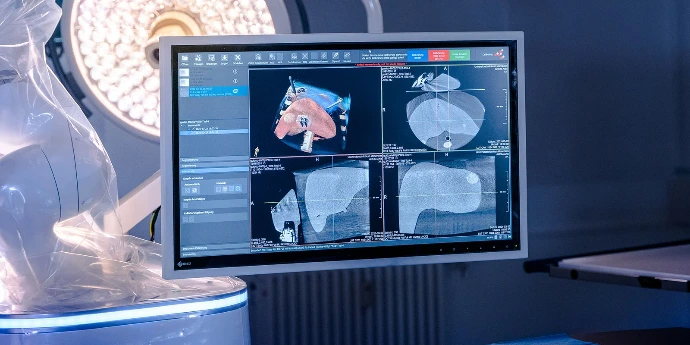
What is
Guidoo
Plain language: what percutaneous biopsies are and why precision matters.
Guidoo guides the needle sleeve to planned puncture site with robot precision.
Key stat: needle placement in <20 minutes from planning scan to control scan.
Transparency: guidoo is under CE approval — current status clearly stated.
Three Clinical Benefits
List and describe the key features of your solution or service.
Speed
<20 min needle placement. High value for multi-needle procedures and extreme insertion angles.
Precision
Imaging-based navigation. Very high path fidelity. Minimises collateral tissue damage.
Simplicity
Robot guides the needle sleeve. Clinician retains full control. High confidence placement.
Evidence-Based Development
Guidoo's development is grounded in peer-reviewed research, independently validated accuracy data and established research partnerships with leading medical engineering institutions.
Milestones Timeline
Guidoo has progressed from research concept to First-in-Human study, with independent recognition from the global robotics industry along the way.
Development
Guidoo was developed by BEC Robotics in collaboration with Fraunhofer MEVIS and MediMesh, combining BEC's robotics engineering expertise with advanced medical image processing and interventional planning technology.

A timeline is a graphical representation on which important events are marked.
First-in-Human Study
A First-in-Human clinical study has been initiated, marking a critical step in Guidoo's pathway toward regulatory approval and clinical adoption. The study evaluates safety and performance in real interventional procedures.

A timeline is a graphical representation on which important events are marked.
KUKA Innovation Award Finalist 2025
Guidoo was selected as a finalist for the KUKA Innovation Award 2025, recognising its contribution to the advancement of robotics in medical applications and its potential for clinical impact.

A timeline is a graphical representation on which important events are marked.
Approval Pathway
BEC is progressing Guidoo through the CE approval pathway under Medical Device Regulation. Current regulatory status is available on request. [CLIENT TO CONFIRM current status and expected timeline].

A timeline is a graphical representation on which important events are marked.
Projects
Check out what's new in our company !
Related Services
List and describe the key features of your solution or service.
Exacure
Modular robotic patient positioning system for BNCT, particle therapy and radiotherapy. CE-marked and clinically installed at leading treatment centres worldwide.
Frequently asked questions
Here are some common questions about our company.
Guidoo is not yet commercially approved for clinical use. It is currently progressing through the CE approval pathway under Medical Device Regulation, and a First-in-Human study has been initiated. BEC provides transparent updates on regulatory status to interested clinical partners. Please contact us for current information.
Guidoo is designed for percutaneous interventional procedures including needle biopsies and ablative treatments. It guides the interventional needle sleeve to the planned puncture site with robot precision, reducing procedure time and the number of iterations required for accurate needle placement.
Guidoo uses imaging-based navigation with needle placement planned from CT imaging data. Compatibility with specific imaging systems in your facility can be assessed by BEC's medical team.
Accuracy data from the published feasibility study (Lautenschläger et al., In Vivo 2023) demonstrates clinically relevant needle placement precision. The system is tested for placement and superimposition accuracy with each installation. Full accuracy data is available in the published reference or on request from BEC.
BEC is progressing Guidoo through the CE approval process under Medical Device Regulation. The timeline depends on regulatory body review timelines and clinical study outcomes. Please contact BEC's medical team for the most current status. [CLIENT TO CONFIRM].
Contact Form
Use this section to boost your company's credibility.
Interested in Guidoo for Your Institution?
Contact BEC's medical team to discuss Guidoo's development status, clinical study participation opportunities, or integration planning for your interventional suite.


